Differences in PEMF Therapy Devices
Power Level
The magnetic energy produced by the various PEMF devices can be as little as that of the Earth’s magnetic field to more than 10,000 times as powerful. The lower power devices are generally used for cellular health and bone healing. The higher power devices are generally used for recovery of trauma from accidents, sports injuries and surgery, as well as for control and improvement of degenerative diseases. Both low power and high power devices help reduce pain, but the higher power devices are more effective in doing so.
Continuous or Pulsed Waveform
Although there are exceptions in both types, most low power PEMF devices have a continuous waveform while most high power PEMF devices have a pulsed waveform.
Shape of Waveform
The continuous waveform PEMF devices can produce a square, a saw tooth, a sine or a custom waveform. The pulsed output PEMF devices usually produce a biphasic short duration pulse.
Control of Pulse Rate
Many low power PEMF devices have preset pulse rate to choose from according to the various manufacturers’ individual theories. Most high power PEMF devices have a user variable control of the pulse rate.
Duration of Treatment
Depending on the power level of the PEMF device, the treatment duration can be from three minutes to hours.
Primary Benefits of PEMF Therapy
Clinical evidence shows that PEMF therapy reduces pain associated with trauma from accidents, sports injuries, surgeries and burns as well as from disease and degeneration. PEMF therapy improves these conditions in many different concurrent ways including mechanical, chemical, electrical and magnetic processes within the cells of the body.
In 1995, Siskin and Walker provided a summary of clinical results on soft tissue damage. They observed no adverse effects and the following positive effects were reported: Reduced pain
- Reduced pain
- Reduced inflammation
- Increased range of motion
- Faster functional recovery
- Reduced muscle loss after surgery
- Increased tensile strength in ligaments
- Faster healing of skin wounds
- Enhanced capillary formation
- Accelerated nerve regeneration
- Reduced tissue necrosis
In the “Beneficial effects of electromagnetic fields”, Bassett C. (Bioelectric Research Center, Columbia University, NY, 1993) applied time-varying pulsed magnetic fields designed to induce voltages similar to those produced normally during the dynamic mechanical deformation of connective tissues in an effort to control cellular function and understand the mechanisms by which PEMF treatment operates and concluded: “As a result, a wide variety of challenging musculoskeletal disorders has been treated successfully over the past two decades... Many of the athermal bioresponses, at the cellular and subcellular levels, have been identified and found appropriate to correct or modify the pathologic processes for which PEMFs have been used… As understanding of mechanisms expands, specific requirements for field energetics are being defined and the range of treatable ills broadened. These include nerve regeneration, wound healing, graft behavior, diabetes, and myocardial and cerebral ischemia (heart attack and stroke), among other conditions. Preliminary data even suggest possible benefits in controlling malignancy”.
Nitric Oxide Production
Many cells in the body produce nitric oxide; however, its production by the vascular endothelium is particularly important in the regulation of blood flow. Abnormal production of nitric oxide, as occurs in different disease states, can adversely affect blood flow and other vascular functions. Nitric oxide is one of the few gaseous signaling molecules known and is additionally exceptional due to the fact that it is a radical gas. It is a key vertebrate biological messenger, playing a role in biological processes.
The March/April 2009 Aesthetic Surgery Journal published a study: “Evidence-Based Use of Pulsed Electromagnetic Field Therapy in Clinical Plastic Surgery” that summarizes the evolution in the understanding of the physiological effects of PEMF therapy on cells and tissues. Studies emerged suggesting that PEMF could modulate the production of growth factors and began to focus on enzyme systems with well-characterized calcium (Ca2+) dependence. By the mid-1990s, researchers were investigating the effects of electrical and PEMF signaling on intracellular Ca2+, specifically the binding of Ca2+ to calmodulin (CaM), using the knowledge that CaM dependent cascades were involved in tissue repair. The most recent studies of the PEMF transduction pathway have concentrated upon the Ca/CaM-dependent nitric oxide cascades, the growth factor cascades involved in tissue healing. It is within this system that the effectiveness of PEMF is now understood to function. PEMFs modulate the calcium-binding kinetics to calmodulin.
Calcium/calmodulin (Ca/CaM) then activates nitric oxide synthase (NOS) in several different isoforms. When injury occurs, large amounts of nitric oxide are produced by long-lived inducible nitric oxide synthase (iNOS). In this cascade, tissue levels of nitric oxide persist and the prolonged presence of this free radical is proinflammatory, which accounts for the leaky blood vessels associated with pain and swelling. In contrast, the endothelial and neuronal nitric oxide synthase isoforms (respectively eNOS and nNOS) produce nitric oxide in short bursts that can immediately relax blood and lymph vessels. These short bursts of nitric oxide also lead to the production of cyclic guano sine monophosphate (cGMP), which in turn drives growth factor production. Interestingly, iNOS is not dependent on CaM, while the constitutive or cNOS (eNOS or nNOS) cascade is dependent on the binding of Ca/CaM. Therapies that could accelerate Ca/CaM binding, therefore, should impact all phases of tissue repair, from initial pain and swelling to blood vessel growth, tissue regeneration, and remodeling.
As shown in the following diagram, this mechanism has been proposed as a working model for PEMF therapeutics.
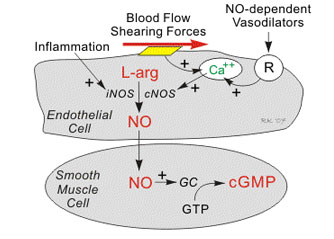
Nitric oxide, known as the 'endothelium-derived relaxing factor', or 'EDRF', is biosynthesized endogenously from L-arginine, oxygen and NADPH by various nitric oxide synthase (NOS) enzymes. Dr. Richard E. Klabunde explains the synthesis of nitric oxide from the amino acid L-arginine by the enzymatic action of nitric oxide synthase (NOS). There are two endothelial forms of NOS: constitutive NOS (cNOS; type III) and inducible NOS (iNOS; type II). In addition to endothelial NOS, there is a neural NOS (nNOS; type I) that serves as a transmitter in the brain and in different nerves of the peripheral nervous system, such as non-adrenergic, non-cholinergic (NANC) autonomic nerves that innervate penile erectile tissues and other specialized tissues in the body to produce vasodilation.
The endothelium (inner lining) of blood vessels uses nitric oxide to signal the surrounding smooth muscle to relax, thus resulting in vasodilation and increasing blood flow. Under normal conditions, nitric oxide is continually being produced by cNOS in the blood vessels. The activity of cNOS is Ca/CaM-dependent and produces vascular relaxation when the endothelium is intact. The activation of the other isoform of endothelial NOS is iNOS is not calcium dependent. Under normal conditions, the activity of iNOS is very low. The activity of iNOS is stimulated during inflammation by bacterial endotoxins or cytokines such as tumor necrosis factor (TNF) and interleukins.
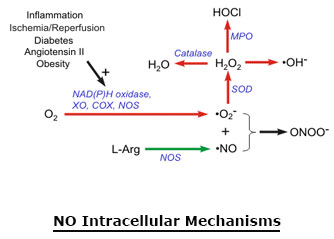 During inflammation, the amount of nitric oxide produced by iNOS may be a 1,000-fold greater than that produced by cNOS. When nitric oxide forms, it is highly reactive (having a lifetime of a few seconds), yet diffuses freely across membranes, primarily because superoxide anion has a high affinity for nitric oxide. Superoxide and its products can have vasoactive activities in addition to their tissue damaging effects; superoxide anion has another property that makes it very important in cardiovascular pathology and pathophysiology. Superoxide anion, with its unpaired electron, very rapidly binds to nitric oxide, which also has an unpaired electron. Because nitric oxide is a very important vasodilator substance, the reaction between superoxide and nitric oxide effectively scavenges nitric oxide thereby reducing its bioavailability. This leads to vasoconstriction, increased platelet-endothelial cell adhesion, platelet aggregation and thrombus formation, increased leukocyte-endothelial cell adhesion, and morphologic changes in blood vessels, such as cell proliferation. Nitric oxide also avidly binds to hemoglobin (in red blood cells) and the enzyme guanylyl cyclase, which is found in vascular smooth muscle cells and most other cells of the body. When nitric oxide is formed by vascular endothelium, it rapidly diffuses into the blood where it binds to hemoglobin and subsequently broken down. It also diffuses into the vascular smooth muscle cells adjacent to the endothelium where it binds to and activates guanylyl cyclase. This enzyme catalyzes the dephosphorylation of GTP to cGMP, which serves as a second messenger for many important cellular functions, particularly for signaling smooth muscle relaxation.
During inflammation, the amount of nitric oxide produced by iNOS may be a 1,000-fold greater than that produced by cNOS. When nitric oxide forms, it is highly reactive (having a lifetime of a few seconds), yet diffuses freely across membranes, primarily because superoxide anion has a high affinity for nitric oxide. Superoxide and its products can have vasoactive activities in addition to their tissue damaging effects; superoxide anion has another property that makes it very important in cardiovascular pathology and pathophysiology. Superoxide anion, with its unpaired electron, very rapidly binds to nitric oxide, which also has an unpaired electron. Because nitric oxide is a very important vasodilator substance, the reaction between superoxide and nitric oxide effectively scavenges nitric oxide thereby reducing its bioavailability. This leads to vasoconstriction, increased platelet-endothelial cell adhesion, platelet aggregation and thrombus formation, increased leukocyte-endothelial cell adhesion, and morphologic changes in blood vessels, such as cell proliferation. Nitric oxide also avidly binds to hemoglobin (in red blood cells) and the enzyme guanylyl cyclase, which is found in vascular smooth muscle cells and most other cells of the body. When nitric oxide is formed by vascular endothelium, it rapidly diffuses into the blood where it binds to hemoglobin and subsequently broken down. It also diffuses into the vascular smooth muscle cells adjacent to the endothelium where it binds to and activates guanylyl cyclase. This enzyme catalyzes the dephosphorylation of GTP to cGMP, which serves as a second messenger for many important cellular functions, particularly for signaling smooth muscle relaxation.
Because of the central role of cGMP in nitric oxide mediated vasodilation, drugs (e.g., Viagra®) that inhibit the breakdown of cGMP (cGMP-dependent phosphodiesterase inhibitors) are used to enhance nitric oxide mediated vasodilation, particularly in penile erectile tissue in the treatment of erectile dysfunction. Increased cGMP also has an important anti-platelet, anti-aggregatory effect. (Cardiovascular Physiology Concepts by Richard E. Klabunde, PhD, published in 2005, www.cvphysiology.com updated in 2008).
In the Discussion in a study entitled “Pulsed Electro-Magnetic Fields Affect Local Factor Production and Connexin 43 Protein Expression in MLO-Y4 Osteocyte-like cells and ROS17/2.8 Osteoblasts like Cells”, Lohman C.H. et al. state: “This study shows that PEMF affects gap junction formation, local production of nitric oxide, TGF-B1 and PGE2. Osteocytes potentially regulate the bone remodeling through signaling molecules like nitric oxide and PGE2 but also through the local release of TGF-B1.”
The above studies demonstrate that PEMF therapy affects many transduction pathways and, in particular the Ca/CaM-dependent nitric oxide cascades. The CaM dependent cascades are involved in tissue repair. By modulating the calcium-binding kinetics to calmodulin (intracellular Ca2+/CaM), the endothelial and neuronal nitric oxide synthase isoforms (respectively eNOS and nNOS) produce nitric oxide in short bursts that can immediately relax blood and lymph vessels. As a highly reactive gaseous molecule, nitric oxide makes an ideal transient paracrine (between adjacent cells) and autocrine (within a single cell) signaling molecule that has direct and indirect vascular action, including the following:
- Direct vasodilation (flow dependent and receptor mediated)
- Indirect vasodilation by inhibiting vasoconstrictor influences
- Anti-thrombotic effect - inhibits platelet adhesion to the vascular endothelium
- Anti-inflammatory effect - inhibits leukocyte adhesion to vascular endothelium; scavenges superoxide anion
- Anti-proliferative effect - inhibits smooth muscle hyperplasia
By increasing the production of nitric oxide when its production is impaired or its bioavailability is reduced, PEMF therapy can successfully help improve conditions and diseases, including those associated with vasoconstriction (e.g., coronary vasospasm, elevated systemic vascular resistance, hypertension), thrombosis due to platelet aggregation and adhesion to vascular endothelium, inflammation due to upregulation of leukocyte and endothelial adhesion molecules, vascular hypertrophy and stenosis, and consequently hypertension, obesity, dyslipidemias (particularly hypercholesterolemia and hypertriglyceridemia), diabetes (both type I and II), heart failure, atherosclerosis, tissue repair and aging.
A recent study on postoperative recovery led to the conclusion that PEMF therapy significantly reduced postoperative pain and narcotic use in the immediate postoperative period by means of a PEMF effect on nitric oxide signaling, which could impact the speed and quality of wound repair (Rohde et al., June 2009, Plastic & Reconstructive Surgery, Columbia, NY).
Nitric oxide is one of the few gaseous signaling molecules and a key vertebrate biological messenger that plays a role in a variety of biological processes. Recent studies uncover how PEMF therapy stimulates and rebalances many of these processes.
The mechanisms by which nitric oxide has been demonstrated to affect the biology of living cells are numerous and include oxidation of iron-containing proteins such as ribonucleotide reductase and aconitase, activation of the soluble guanylate cyclase, a single transmembrane protein, ADP (adenosine di-phosphate) ribosylation of proteins, a process of protein modification involved in cell signaling and the control of many cell processes including DNA repair, protein sulfhydryl group nitrosylation, another protein modification process, and iron regulatory factor activation. Having a lifetime of a few seconds, nitric oxide is highly reactive and diffuses freely across cell membranes. These attributes make nitric oxide an ideal transient paracrine (between adjacent cells) and autocrine (within a single cell) signaling molecule. PEMF therapy is proven to effectively stimulate paracrine and autocrine communication.
Nitric oxide is also generated by phagocytes (monocytes, macrophages, and neutrophils) and, as such, is part of the human immune response. Nitric oxide has been demonstrated to activate NF-?B in peripheral blood mononuclear cells, an important protein complex that controls the transcription of DNA and a transcription factor in iNOS gene expression in response to inflammation.
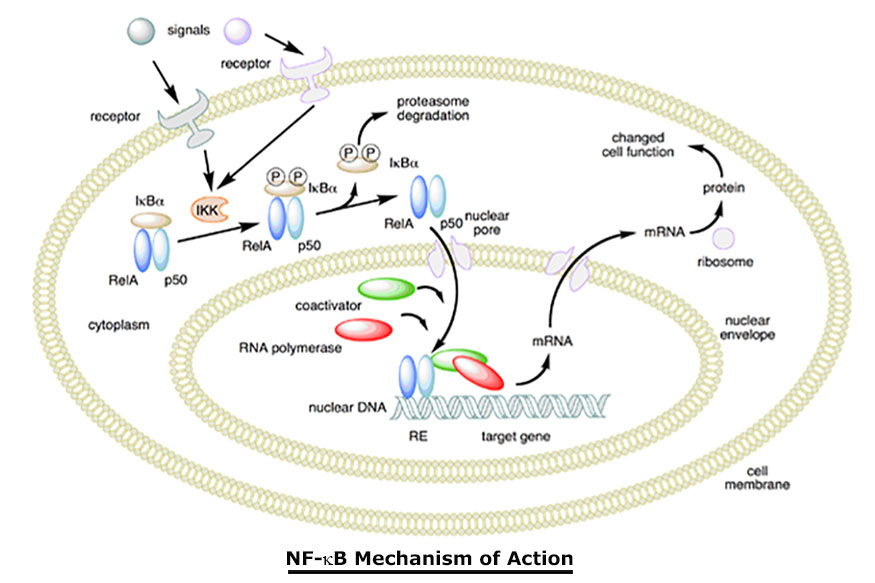
Nitric oxide plays a key role in regulating the immune response to infection and is implicated in processes of synaptic plasticity and memory (see diagram above). The endothelium (inner lining) of blood vessels uses nitric oxide to signal the surrounding smooth muscle to relax, thus resulting in vasodilatation and increasing blood flow. As blood flow increases, so does the oxygen intake. PEMF therapy has proven to increase blood flow and provide muscle relaxation maybe with better oxygenation of the muscle tissue.
During the last 100 years, theories of pain mechanism have evolved from specificity and summation models to the popular Gate Control Theory. The latter pain theory, proposed by Melzack/Wall/Casey (Wall and Melzack, 1989) has become the most important development in the field of pain management. Pain perception is no longer a straightforward afferent transmission of pain signal.
In biology, signal transduction is a mechanism that converts a mechanical or chemical stimulus to a cell into a specific cellular response.
Dynamics of Pain and PEMF Therapy
For most individuals, aside from the multiple benefits of the therapy, one of the most relevant effects of PEMF therapy is the improvement of painful conditions regardless of their origin. Pain mechanisms are complex and have peripheral and central nervous system aspects.
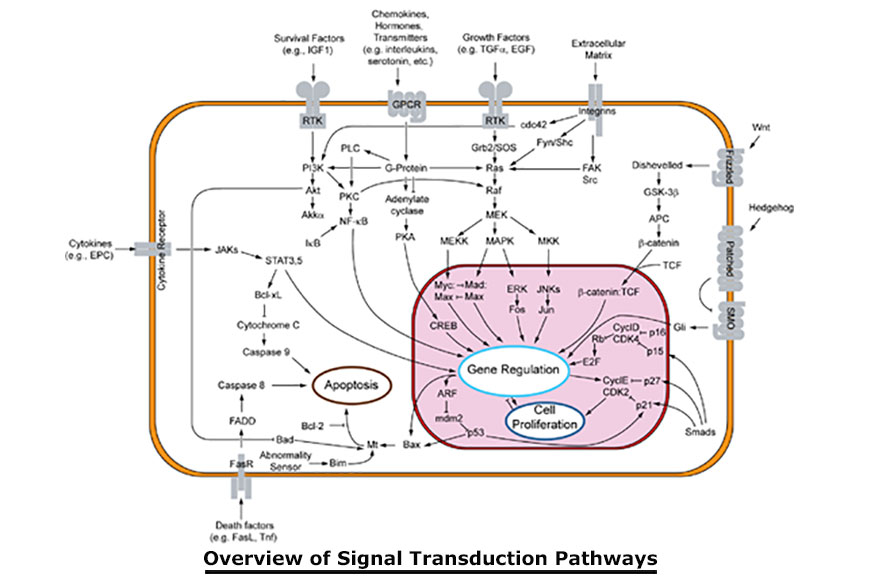
Signal transduction starts with a signal to a receptor, and ends with a change in cell behavior. Transmembrane receptors move across the cell membrane, with half of the receptor outside the cell and the other half inside the cell. The signal, such as a chemical signal, binds to the outer half of the receptor, which changes its shape and conveys another signal inside the cell. Sometimes there is a long cascade of signals, one after the other. Eventually, the signal creates a change in the cell, either in the DNA of the nucleus or the cytoplasm outside the nucleus. In the chronic pain state, pain signal generation can actually occur in the central nervous system without peripheral noxious stimulation. In pain management, modulation of the pain signal transmission is a far better choice than neural destruction, and this can be achieved with PEMF. Scientific evidence shows that acute persistent pain eventually sensitizes wide dynamic neurons in the dorsal horn of the spinal cord, the wind-up phenomenon, constituting the basis of developing chronic pain syndromes (Kristensen, 1992). Persistent and excessive pain has no biological good or necessary function. It is actually harmful to our well-being. Therefore, pain needs to be treated as early and as completely as possible and not to be left alone (Adams et al. 1997).
The primary symptom in most patients with disorders affecting the soft tissue is pain. In many patients, daily activities are limited as inflammation causes pain and, with it, a restriction of the range of movements. Causes of soft tissue pain can be depicted as musculo-skeletal, neurologic, vascular, and referred visceral-somatic or articular (Cailliet, 1991). Early reports of applying electrical current to treat pain date back to before 1800 (Ersek, 1981).
PEMF therapy has successfully been used for the control of pain associated with rotator cuff tendinitis, multiple sclerosis, carpal tunnel syndrome, and peri-arthritis (Battisti et al., 1998; Lecaire et al., 1991). An improvement was observed in 93% of patients suffering from carpal tunnel pain and in 83% in cases of rotator cuff tendinitis. PEMF therapy was also used for treatment of migraine, chronic pelvic pain, neck pain, and whiplash injuries (Rosch et al., 2004).
In a March, 2003 publication on Pain Management with PEMF Treatment, Dr. William Pawluk explains:”Magnetic fields affect pain perception in many different ways. These actions are both direct and indirect. Direct effects of magnetic fields are: neuron firing, calcium ion movement, membrane potentials, endorphin levels, nitric oxide, dopamine levels, acupuncture actions and nerve regeneration. Indirect benefits of magnetic fields on physiologic function are on: circulation, muscle, edema, tissue oxygen, inflammation, healing, prostaglandins, cellular metabolism and cell energy levels… Short-term effects are thought due to a decrease in cortisol and noradrenaline, and an increase in serotonin, endorphins and enkephalins. Longer term effects may be due to CNS and/or peripheral nervous system biochemical and neuronal effects in which correction of pain messages occur; and the pain is not just masked as in the case of medication”.
PEMF Therapy Reduces Pain
Many studies have demonstrated the positive effects of PEMF therapy on patients with pain, even as opposed to receiving traditional treatment as well as against a placebo group getting no treatment. Some studies focused on the rapid, short-term relief while others demonstrate the long-term effects. The effectiveness of PEMF therapy has been demonstrated in a wide variety of painful conditions.
In a study entitled: “Double-blind, placebo-controlled study on the treatment of migraine with PEMF”, Sherman et al. (Orthopedic Surgery Service, Madigan Army Medical Center, Tacoma, WA, USA) evaluated 42 subjects who met the International Headache Society's criteria. During the first month of follow-up with exposure to PEMF, 73% of those receiving actual exposure, reported decreased headaches with 45% a substantial decrease and 14% an excellent decrease. Ten of the 22 subjects who had received actual exposure received two additional weeks of actual exposure, after their initial month. All showed decreased headache activity with 50% a substantial decrease and 38% an excellent decrease. Sherman R. et.al concluded that exposure to PEMF for at least 3 weeks is an effective, short-term intervention for migraine.
Jorgensen et al. (1994 International Pain Research Institute, Los Angeles, CA, USA) studied the effects of PEMF on tissue trauma and concluded: “Unusually effective and long-lasting relief of pelvic pain of gynecological origin has been obtained consistently by short exposures of affected areas to the application of a magnetic induction device. Treatments are short, fasting-acting, economical, and in many instances have obviated surgery”. Patients with typical cases such as dysmenorrhoea, endometriosis, ruptured ovarian cyst, acute lower urinary tract infection, post-operative hematoma, and persistent dyspareunia who had not received analgesic medication were treated with pulsed magnetic field treatment and evaluated. The results showed that 90% of the patients experienced marked, even dramatic relief, while 10% reported less than complete pain.
Hedén P., Pilla AA. (2008 Department of Plastic Surgery, Stockholm, Sweden) studied the Effects of pulsed electro-magnetic fields on postoperative pain in breast augmentation patients. She notes: “Postoperative pain may be experienced after breast augmentation surgery despite advances in surgical techniques, which minimize trauma. The use of pharmacological analgesics and narcotics may have undesirable side effects that can add to patient morbidity”. This study was undertaken to determine if PEMF could provide pain control after breast augmentation. Postoperative pain data were obtained and showed that pain had decreased in the treated patient group by nearly a factor of three times that for the control group. Patient use of postoperative pain medication correspondingly also decreased nearly three times faster in the active versus the sham groups. Hedén P and Pilla AA concluded: “Pulsed electro-magnetic field therapy, adjunctive to standard of care, can provide pain control with a noninvasive modality and reduce morbidity due to pain medication after breast augmentation surgery”.
The Clinical Rheumatology Journal, volume 26-1, January 2007 (Springer London) reported on the Effectiveness of PEMF therapy in lateral epicondylitis by Kaan Uzunca, Murat Birtane and Nurettin Tastekin (Trakya University Medical Faculty Physical Medicine and Rehabilitation Department, Edirne, Turkey): “We aimed to investigate the efficacy of PEMF in lateral epicondylitis comparing the modality with sham PEMF and local steroid injection”. Patients with lateral epicondylitis were randomly and equally distributed into three groups. One group received PEMF, another sham PEMF, and the third group a corticosteroid + anesthetic agent injection. Pain levels during rest, activity, nighttime, resisted wrist dorsiflexion, and forearm supination were investigated with visual analog scale (VAS). Pain threshold on elbow was determined with an Algometer. All patients were evaluated before treatment, at the third week and the third month. Pain levels were significantly lower in the group treated with the local steroid at the third week but the group treated with PEMF had lower pain during rest, activity and nighttime than the group receiving steroids at the third month.
Lau (School of Medicine, Loma University, USA) reported on the application of PEMF therapy to the problems of diabetic retinopathy. Patients were treated over a 6-week period, 76% of the patients had a reduction in the level of numbness and tingling. All patients had a reduction of pain, with 66% reporting that they were totally pain-free.
Sanseverino et al. (1999, Universita di Bologna, Italy) studied the therapeutic effects of PEMF on joint diseases, in chronic and acute conditions of more than 3,000 patients over a period of 11 years. Follow-up was pursued as constantly as possible. Pain control, recovery of joint mobility and maintenance of the improved conditions represented the parameters for judging the results as good or poor. The chi-square test was applied in order to evaluate the probability that the results are not casual. A general average value of 78.8% of good results and 21.2% of poor results was obtained. The high percentage of good results obtained and the absolute absence of both negative results and undesired side-effects led to the conclusion that PEMF treatment is an excellent physical therapy in cases of joint diseases. A hypothesis is advanced that external magnetic fields influence transmembrane ionic activity.
In a 2008 randomized clinical trial to determine if a physics-based combination of simultaneous static and time-varying dynamic magnetic field stimulation to the wrist can reduce subjective neuropathic pain and influence objective electrophysiologic parameters of patients with carpal tunnel syndrome, Weintraub et al. report:” PEMF exposure in refractory carpal tunnel syndrome provides statistically significant short- and long-term pain reduction and mild improvement in objective neuronal functions.
In a 2009 evidence-based analysis on the use of PEMF therapy in clinical plastic surgery, Strauch et al. (Einstein College of Medicine, Bronx, NY, USA) explain:” Our objective was to review the major scientific breakthroughs and current understanding of the mechanism of action of PEMF therapy… The results show that PEMF therapy has been used successfully in the management of postsurgical pain and edema, the treatment of chronic wounds, and in facilitating vasodilatation and angiogenesis… with no known side effects for the adjunctive, noninvasive, nonpharmacologic management of postoperative pain and edema… Given the recent rapid advances in development of PEMF devices what has been of most significance to the plastic surgeon is the laboratory and clinical confirmation of decreased pain and swelling following injury or surgery”.
Because of the interaction between the biological systems and natural magnetic fields, PEMFs can affect pain perception in many different ways.
PEMF Therapy Blocks Pain
PEMF therapy has shown to be effective at reducing pain both in the short-term and in the long-term. The ways by which PEMF therapy relieves pain include pain blocking, decreased inflammation, increased cellular flexibility, increased blood and fluids circulation, and increased tissue oxygenation.
The trans-membrane potential, (“TMP”) is the voltage difference (or electrical potential difference) between the interior and exterior of a cell. An electrochemical gradient results from a spatial variation of both an electrical potential and a chemical concentration across a membrane. Both components are often due to ion gradients, particularly proton gradients, and the result is a type of potential energy available for cellular metabolism. This can be calculated as a thermodynamic measure, an electrochemical potential that combines the concepts of energy stored in the form of chemical potential, which accounts for an ion's concentration gradient across a cellular membrane, and electrostatics, which accounts for an ion's tendency to move relative to the TMP.
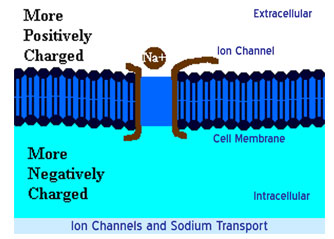
Differences in concentration of ions on opposite sides of a cellular membrane produce the TMP. The largest contributions usually come from sodium (Na+) and chloride (Cl–) ions which have high concentrations in the extracellular region, and potassium (K+) ions, which along with large protein anions have high concentrations in the intracellular region. Opening or closing of ion channels for ion transport (Na+, Ca2+, K+, Cl-) in and out of cells at one point in the membrane produces a local change in the TMP, which causes an electric current to flow rapidly to other points in the membrane that occurs with the movement of electrons.
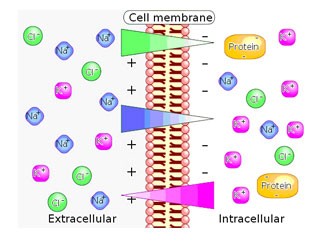
In electrically excitable cells such as neurons, the TMP is used for transmitting signals from one part of a cell to another. In non-excitable cells, and in excitable cells in their baseline states, the TMP is held at a relatively stable value, called the resting potential. For neurons, typical values of the resting potential range from -70 to -80 mV (mill Volts); that is, the interior of a cell has a negative baseline voltage. Each axon has its characteristic resting potential voltage and in each case the inside is negative relative to the outside.
Opening and closing of ion channels can induce a departure from the resting potential, called a depolarization if the interior voltage rises (say from -70 mV to -65 mV), or a hyper polarization if the interior voltage becomes more negative (for example, changing from -70 mV to -80 mV).
In excitable cells, a sufficiently large depolarization can evoke a short-lasting all-or-nothing event called an action potential, in which the TMP very rapidly undergoes a large change, often reversing its sign. Special types of voltage-dependent ion channels that generate action potentials but remain closed at the resting TMP can be induced to open by a small depolarization.
 In a lecture on Pain Reduction, Dr. D. Laycock, Ph.D. Med. Eng. MBES, MIPEM, B.Ed., inspired by the works of Adams et al. (1997) explains how PEMF therapy affects pain transmission at the levels of the neurons. “It is necessary to understand the mechanism of pain transmission to understand how pain blocking can take place with PEMF therapy. Pain is transmitted along the nerve cells by an electric signal. This signal encounters synaptic gaps at intervals. The pain signals are transmitted along nerve cells to pre-synaptic terminals. At these terminals, channels in the cell alter due to a movement of ions. The TMP changes, causing the release of a chemical transmitter from a synaptic vesicle contained within the membrane. The pain signal is chemically transferred across the synaptic gap to chemical receptors on the post-synaptic nerve cell. This all happens in about 1/2000th of a second, as the synaptic gap is only 20 to 50 nm (nano meters) wide. As the pain signal, in chemical form, approaches the post-synaptic cell, the membrane changes and the signal is transferred. During quiescent times, cells possess a small charge of about –70mV between the inner and outer membranes. When a pain signal arrives, it temporarily depolarizes the nociceptive cell and raises the cell TMP to +30mV. This increase is sufficient to open channels in the cell membrane allowing the exchange of the sodium (Na+) and potassium (K+) ions.
In a lecture on Pain Reduction, Dr. D. Laycock, Ph.D. Med. Eng. MBES, MIPEM, B.Ed., inspired by the works of Adams et al. (1997) explains how PEMF therapy affects pain transmission at the levels of the neurons. “It is necessary to understand the mechanism of pain transmission to understand how pain blocking can take place with PEMF therapy. Pain is transmitted along the nerve cells by an electric signal. This signal encounters synaptic gaps at intervals. The pain signals are transmitted along nerve cells to pre-synaptic terminals. At these terminals, channels in the cell alter due to a movement of ions. The TMP changes, causing the release of a chemical transmitter from a synaptic vesicle contained within the membrane. The pain signal is chemically transferred across the synaptic gap to chemical receptors on the post-synaptic nerve cell. This all happens in about 1/2000th of a second, as the synaptic gap is only 20 to 50 nm (nano meters) wide. As the pain signal, in chemical form, approaches the post-synaptic cell, the membrane changes and the signal is transferred. During quiescent times, cells possess a small charge of about –70mV between the inner and outer membranes. When a pain signal arrives, it temporarily depolarizes the nociceptive cell and raises the cell TMP to +30mV. This increase is sufficient to open channels in the cell membrane allowing the exchange of the sodium (Na+) and potassium (K+) ions.
When an action potential begins, the channels that allow crossing of the Na+ ions open up. When the Na+ channels open, the depolarization occurs, the Na+ rushes in because both of the greater concentration of Na+ on the outside and the more positive voltage on the outside of the axon. The flow of positively charged ions into the axon leads the axon to become positively charged relative to the outside. With each positively charged Na+ ion that enters the axon, another positive charge is inside and one fewer negative charge is outside the axon. Thus, together the inside grows increasingly more positive and the relative concentration of Na+ inside the axon relative to outside the axon grows greater. This initial phase of the action potential is called the depolarization phase. Now as the depolarization phase progresses, the status of the two physical forces that have been discussed changes. At the end of the depolarization phase, the voltage of the inside of the axon relative to the outside is positive and the relative concentration of Na+ ions inside the axon is greater than at the beginning of the action potential. The inside of the axon becomes sufficiently positive, about +30 mV as an average value, the Na+ channels close. This closing of the Na+ channels will greatly limit the ability of Na+ ions to enter the axon. In addition to the Na+ channels closing, the potassium (K+) channels open. Now K+ ions are free to cross the channels and now leave the axon due both to the greater concentration of K+ on the inside and the reversed voltage levels. The action potential is therefore not the movement of voltage or ions but the flow of these ion channels opening and closing moving down the axon.
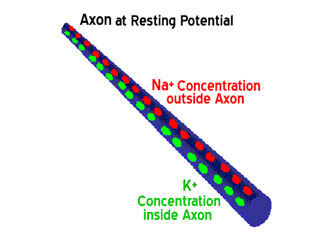
This movement of the ion channels explains why the action potential is transferred slowly relative to the normal flow of electricity. The normal flow electricity is the flow of electrons in an electrical field and the electrons travel at the speed of light while the movement of these ion channels opening and closing is considerably slower. These are mechanical movements that cannot move as fast as the speed of light.
The exchange of the sodium (NA+) and potassium (K+) ions then triggers exocytosis of neurotransmitters via synaptic vesicles. These neurotransmitters diffuse into the synaptic gap. Once this process has occurred, the cell depolarizes back to its previous level of –70mV.
Research by Warnke established that the application of PEMF therapy has an effect on the quiescent potential of the neuronal synaptic membrane (Warnke, 1983; Warnke et al. 1997). It suggested that the effect is to lower the potential to a hyperpolarized level of –90mV. “When a pain signal is received, the TMP has to be raised again in order to fire an action potential via neurotransmitters but it only achieves to raise the cell TMP to an approximate +10mV. This potential is well below the threshold of +30mV necessary to release the relevant neurotransmitters into the synaptic cleft and the pain signal is effectively blocked”.
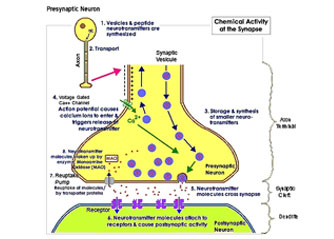
By causing a hyperpolarized state at the neuronal membrane, PEMF therapy effectively blocks pain as it prevents the threshold necessary to transmit the pain signal to be reached. In the same way, PEMF therapy effectively increases the TMP of damaged cells thus allowing them to recover their functions, heal and improve their metabolism.
The Encyclopedia of Nursing and Allied Health define the use of “Electrotherapy” for pain relief as effective to manage both acute and chronic pain.
In the “Gate Model” of pain, the neural fibers that carry the signal for pain and those that carry the signal for proprioception (body and limb position) are mediated through the same central junction. Because signal transmission along pain fibers is slower than transmission along proprioception fibers, the Gate model suggests that intense stimulation of proprioception fibers can block the slower-moving pain signals.
PEMF Therapy Reduces Inflammation
Several factors may contribute to inflammation including injury, tissue damage, a poor localized circulation with the formation of edema. Inflammation causes pain. Swelling and bruising is an inflammation and discoloration of soft tissue caused by an impact injury or trauma. It can also result from surgery.
Tissue cells are inherently like tiny electrically charged machines. When a cell is traumatized, the cell’s electrical charge is diminished; this causes normal cell functions and operations to shut down. Cells that are scarred or fibrotic with adhesions have a TMP charge of approximately -15 mV, degenerative or immune-compromised cells average -30 mV, both low TMPs. With the raised TMP, the body releases chemical signals that cause inflammation swelling and bruising resulting in pain and inhibiting the cell communication pathways necessary for healing to begin. Numerous clinical studies have demonstrated that PEMF therapy has been successful in reducing inflammation. PEMF therapy treats the cellular source of swelling by recharging the cells with a mild electromagnetic current. This stops the release of pain and inflammatory mediators, reduces inflammatory fluids and allows an increase in blood flow, therefore increased oxygen intake, to help the cells heal faster with less swelling, pain and bruising.
The effect of wound healing electromagnetic fields on inflammatory cytokine gene expression in rats was studied by Jasti et al. in 2001 who state: “Inflammation is characterized by massive infiltration of T lymphocytes, neutrophils and macrophages into the damaged tissue. These inflammatory cells produce a variety of cytokines, which are the cellular regulators of inflammation”. In a study on Low Frequency PEMF—a viable alternative therapy for arthritis published in 2009, Ganesan et al. (Department of Biotechnology, Chennai, India) declare: “PEMF for arthritis cure has conclusively shown that PEMF not only alleviates the pain in the arthritis condition but it also affords chondroprotection, exerts anti-inflammatory action and helps in bone remodeling, and this could be developed as a viable alternative for arthritis therapy”.
Damaged cells are also energy deficient; thus they have low oxygen levels, high in sodium levels, and have a faltered electrochemical gradient. By inducing a mild electrical current into damaged cells, PEMF therapy slows or stops the release of pain and inflammatory mediators, increases blood flow, and re-establishes normal cell interaction. PEMF stimulates and restores the electrochemical gradient, the cell starts pumping sodium out, potassium enters the cell, the swelling resolves, oxygen starts flowing back in, and pain improves. Due to the density of the cell tissue, change requires stronger pulsed magnetic fields to be able to restore the healthy TMP to its optimal -70 mV.
Several factors influence tissue inflammation and the processes by which PEMF therapy operates to reduce inflammation include complex mechanical, chemical, electrical and magnetic processes along with increased circulation, oxygenation and cellular activity. With reduced inflammation, pain decreases and faster tissue healing occurs.
The Elsevier Journal of Biomedicine & Pharmacotherapy (2005) publication: “Effects of pulsed electromagnetic fields on articular hyaline cartilage: review of experimental and clinical studies by M. Fini. G. Giavaresi, A. Carpi, A. Nicolini, S. Setti, R. Giardino (Experimental Surgery Department, Research Institute Codivilla-Putti-Rizzoli, Orthopedic Institute, via di Barbiano 1/10, 40136 Bologna, Italy, Department of Reproduction and Aging, University of Pisa, Pisa, Italy, Department of Internal Medicine, University of Pisa, Pisa, Italy, igea SRL, Carpi, Modena, Italy) states: “Newer concepts on osteoarthritis (OA) pathogenesis are related to the role of inflammation that is now well accepted as a feature in OA. Synovitis is common in advanced age involving infiltration of activated B cells and T lymphocytes and the expression of pro-inflammatory cytokines and chemokines is observed in patients with OA in the joints of OA patients and animals. With regards to this, IL-1?, TNF?, IL-6, IL-18, IL-17 and leukemia inhibitory factor (LIF) appear to be more relevant to the disease. These catabolic cytokines lead to the destruction of joint tissue by stimulating cartilage PG resorption, MMP synthesis and nitric oxide production. The purine base adenosine has been shown to limit inflammation through receptor (i.e. A2a)-mediated regulation and suppressing pro-inflammatory cytokines synthesis (TNF?, IL-8, IL-2, IL-6). Adenosine has been reported to reduce inflammation and swelling in several in vivo models of inflammation and also in adjuvant-induced and septic arthritis in animals. So, a therapy combining an anabolic effect on chondrocytes, a catabolic cytokine blockage, a stimulatory effect on anabolic cytokine production and one that is able to counteract the inflammatory process would be extremely useful for OA treatment. In vitro studies showed that chondrocyte proliferation and matrix synthesis are significantly enhanced by PEMF stimulation, when investigating also the conditions affecting the PEMF action. A part the importance of physical properties of the fields used (intensity, frequency, impulse amplitude, etc.) and the exposure time, the availability of growth factors, environmental constrictions and the maintenance of the native–cell matrix interactions seem to be fundamental in driving the PEMF-induced stimulation. In particular, the interaction between cell membrane receptors and mitogens seems to be one of the molecular events affected by PEMFs. These data are in agreement with results of in vivo studies with a decalcified bone matrix induced endochondral ossification model and showing that the stimulation of TGF-?1 may be a mechanism through which PEMFs affect complex tissue behavior and through which the effects of PEMFs may be amplified. In addition, PEMFs are reported to up-regulate mRNA levels for, and protein synthesis of, growth factors resulting in the synthesis of ECM proteins and acceleration of tissue repair. As far as inflammation is concerned, IL-1? is present in high amounts in OA cartilage and is considered to be one of the main catabolic factors involved in the cartilage matrix degradation associated with OA. As previously mentioned, PEMFs in vitro were able to counterbalance efficiently the cartilage degradation induced by the catabolic cytokine”.
As cited above, many studies lead to the conclusion that PEMF therapy is effective and reduces inflammation.
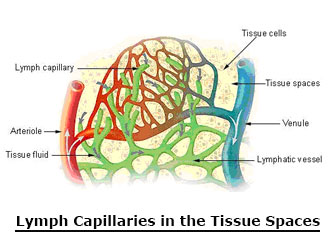
PEMF Therapy Increases Blood and Lymphatic Circluation
The arterial and venal blood vessels are intimately associated with the lymphatic system.. As the blood and lymphatic vessels bring oxygen and nutrients to the cells and remove their waste products, they are nourishing and detoxifying the cells, tissues and body.
As PEMF therapy mechanically stimulates blood vessels and blood flow, the blood vessels pump blood and oxygen into the cells. Simultaneously, PEMF therapy mechanically stimulates the lymphatic vessels and waste products are hauled away from the cells more efficiently. PEMF therapy supports immune health by mechanically stimulating lymphatic drainage and blood flow.
In June 2004, The Faseb Journal states: “PEMF therapy has been shown to be clinically beneficial in repairing bones and other tissues, but the mechanism in action is unclear. The results of a study done at the New York University Medical Center (Institute of Reconstructive Plastic Surgery, NY, NY, USA) demonstrates that electro-magnetic fields increased angiogenesis, the growth of new blood vessels, in vitro and in vivo through the endothelial release of FGF-2, fibroblast growth factor-2. The delivery of PEMF therapy in low doses identical to that currently in clinical use significantly increased endothelial cell proliferation
and tubulization, which are both important processes for vessel formation.
 The ability of PEMF to increase cell proliferation was unique to endothelial cells, which seemed to be the primary target of PEMF stimulation, releasing a protein in a paracrine fashion (or signaling to adjacent cells and other types of cells) to induce changes in neighboring cells and tissues. Since direct stimulation did not produce significant changes in osteoblast proliferation, the ability of PEMF therapy to enhance the healing of complicated fractures is likely the result of increased vascularity rather than a direct effect on osteogenesis as previously believed. The coordinated release of FGF-2 suggests that PEMF therapy may facilitate healing by augmenting the interaction between osteogenesis and blood vessel growth. As such, PEMF therapy may offer distinct advantages as a non-invasive and targeted modality that is able to release several growth factors to achieve therapeutic angiogenesis. The fibroblast and endothelial cells are made to go embryonic due to drastic changes in ionic concentrations in the cells’ cytoplasm and therefore the cells’ nuclei. These ionic concentrations react with the cell DNA opening up some gene sets and closing down others. It is apparently the rapid onset of a strong-pulsed electric field generated by the pulsed magnetic field, which causes some cell ion gate types to open and be force fed ions by the same electric field”.
The ability of PEMF to increase cell proliferation was unique to endothelial cells, which seemed to be the primary target of PEMF stimulation, releasing a protein in a paracrine fashion (or signaling to adjacent cells and other types of cells) to induce changes in neighboring cells and tissues. Since direct stimulation did not produce significant changes in osteoblast proliferation, the ability of PEMF therapy to enhance the healing of complicated fractures is likely the result of increased vascularity rather than a direct effect on osteogenesis as previously believed. The coordinated release of FGF-2 suggests that PEMF therapy may facilitate healing by augmenting the interaction between osteogenesis and blood vessel growth. As such, PEMF therapy may offer distinct advantages as a non-invasive and targeted modality that is able to release several growth factors to achieve therapeutic angiogenesis. The fibroblast and endothelial cells are made to go embryonic due to drastic changes in ionic concentrations in the cells’ cytoplasm and therefore the cells’ nuclei. These ionic concentrations react with the cell DNA opening up some gene sets and closing down others. It is apparently the rapid onset of a strong-pulsed electric field generated by the pulsed magnetic field, which causes some cell ion gate types to open and be force fed ions by the same electric field”.
As demonstrated in the following study entitled: “Impulse magnetic-field therapy for erectile dysfunction: a double-blind, placebo-controlled study”, increased microcirculation leads to improvements in macro-circulation. The study by Pelka et al. (Universitat der Bundeswehr Munchen, Munich, Germany) assessed the efficacy of three weeks of PEMF therapy for erectile dysfunction. In the active-treatment group, all efficacy endpoints were significantly improved at study end with 80% reporting increases in intensity and duration of erection, frequency of genital warmth, and general well-being. In contrast, only 30% of the placebo group noted some improvement in their sexual activity; 70% had no change. No side effects were reported.
PEMF therapy has proven efficacious at increasing the flow of ions and nutrients into the cells and at stimulating blood and interstitial fluid circulation. With increased lymphatic drainage and blood flow, cells receive more oxygen and nutrients, and eliminate toxins faster. Cells are therefore able to function better and tissues repair themselves more efficiently. Through the same processes, vital organs such as the liver, kidneys and colon are able to rid themselves of impurities thus detoxifying the body and allowing better organ functionality.
PEMF Therapy Increases Cellular Membrane Permeability
As early as 1940, it was suggested that magnetic fields affect the TMP and the flow of ions in and out of the cells and might therefore influence cellular membrane permeability. It has since been established that magnetic fields can influence ATP (Adenosine Tri-phosphate) production; increase the supply of oxygen and nutrients via the vascular and lymphatic systems; improve the removal of waste via the lymphatic system; and help re-balance the distribution of ions across the cell membrane. Healthy cells in tissue have a voltage difference between the inner and outer membrane referred to as the membrane resting potential that ranges from -70 to -80 mV. This causes a steady flow of ions through its voltage-dependant ion channels. In a damaged cell, the potential is raised and an increased sodium inflow occurs. As a result, interstitial fluid is attracted to the inner cellular space, resulting in swelling and edema. The application of PEMF to damaged cells accelerates the re-establishment of normal potentials (Sanseverino, 1999) increasing the rate of healing and reducing swelling. In biology, depolarization is a change in a cell's TMP, making it more positive or less negative. In neurons and some other cells, a large enough depolarization may result in an action potential. Hyper polarization is the opposite of depolarization, and inhibits the rise of an action potential.
If a cell has a resting potential of -70mV and the membrane potential rises to -50mV, then the cell has been depolarized. Depolarization is often caused by influx of cations, e.g. Na+ through Na+ channels, or Ca2+ through Ca2+ channels. On the other hand, efflux of K+ through K+ channels inhibits depolarization, as does influx of Cl– (an anion) through Cl– channels. If a cell has K+ or Cl– currents at rest, then inhibition of those currents will also result in a depolarization.
As the magnetic field created fluctuates, it induces an electron flow or a current in one direction through the living tissue. As electrons always flow from a negative (cathode) to a positive (anode) potential, when the magnetic field vanishes, the direction of the electron flow is reversed. Therefore such induced polarized currents stimulate the exchange of ions across the cell membrane. As the electro-magnetic field pulses temporarily hyperpolarize and depolarize the membrane, the ion channels open and close allowing a more efficient ion exchange, as with the sodium-potassium (Na+, K+) pump, thus increasing cellular oxygenation and nutrition as sodium export stimulates several secondary active transporters.
Increased cellular membrane permeability thus allows better oxygen and nutrient uptake by the cells as well as increased toxin release from the cells. Cells are therefore better equipped to repair themselves, regenerate, and improve all their cellular functions and genesis.
Another benefit of increased cellular membrane permeability is that of the increased absorption of chemicals available in the blood and lymphatic fluids surrounding a tissue area treated. As demonstrated by many medical studies, this phenomenon can be quite significant in the treatment cancerous tumors and localized cancers.
Cancer and Cellular Proliferation
As with all forms of new therapy, the incidence of negative side effects is a major concern. Since PEMF therapy is known to stimulate cellular genesis, a major concern in patients with cancer or cancerous tumors would be that PEMF therapy might stimulate the development of cancerous cells and tumors.
In study entitled: “Effect of Pulsing Electromagnetic Fields on Invasion Ability in Osteosarcoma Cells.” Journal of the Japanese Bio-Electrical Research Society in 1999, Kobayashi Kenji et al. (Nagoya University, Japan) declare: “We have been trying to apply pulsing electromagnetic fields (PEMF) to chemotherapy in bone and soft tissue sarcomas. Before clinical study, we must know by in vitro study whether PEMF affects the cause of malignancy or not. Therefore we examined the effect of PEMF on invasion ability which correlates well with metastasis. The invasion through artificial basement membrane was quantified by invasion assay in a Boyden chamber. In all experiments, there was no difference between PEMF treated group and control. Thus we conclude that invasion ability is not affected by PEMF exposure in vitro”.
In a study published in the Journal of Bioelectromagnetics in April 2011, Zhang D, Pan X, Ohno S, Osuga T, Sawada S, Sato K. of the Department of Orthopedic Surgery, Aichi Medical University School of Medicine, Nagakute, Aich, Japan and the Department of Orthopaedic Surgery, the First Hospital, Jilin University, Changchun, China, studied the effects of pulsed electromagnetic fields on expression of cell adhesion molecules and matrix metalloproteinase in osteosarcoma cell lines. “Pulsed electromagnetic fields (PEMF) could enhance the cytocidal effects of chemotherapeutic drugs on malignant tumor cell lines, but metastasis effects of PEMF on tumor cells have not been investigated”. The study investigated the effects PEMF exposure on the expression levels of some metastasis-related molecules, including integrin a subunits (a1, a2, a3, a4, a5, a6, av), integrin ß subunits (ß1, ß2, ß3, ß4), CD44, and matrix metalloproteinase-2/9 (MMP-2/9) in four human osteosarcoma cell lines. They conclude: “PEMF exposure has no effect on the expression of some molecules that are associated with tumor cell invasion and metastasis, and therefore suggest that PEMF exposure may be safely applied to chemotherapy for osteosarcoma”.
Cancer and the Immune System
An increase in cellular membrane permeability that promotes more efficient cellular function may also stimulate the immune response to cancer and help the immune system prevent its evolution.
In a study entitled “Immunocorrective effect of alternating magnetic field in the postoperative period in malignant bladder cancer” published in the 2001 Vopr Onkol. Journal, Zlatnik EIu et al. of the Research Institute of Oncology, Ministry of Health of the RF, Rostov-on-Don, Russia state: ”The study deals with immune status of patients operated for bladder cancer and exposed postoperatively to alternating magnetic field (MF) (hypothalamus and operative field). MF application was followed by higher T- and B-lymphocyte and CD4+, CD16+ cell levels as well as enhanced T-cell activity; no postoperative complications were registered and tumor relapse rates were relatively low. The effect was likely to be due to antistressor influence of MF. The procedure may substitute drug therapy for immunocorrection and to avoid recurrence of bladder cancer”.
Cancer and Drug Absorption
Many In Vitro studies on drug resistant human cell lines and In Vivo studies, mostly on rodents, prove the enhanced effects of chemotherapy with the use of PEMF as adjunct therapy.
In December 2002, the journal Bioelectromagnetics published a study on the “Influence of 1 and 25 Hz, 1.5 mT magnetic fields on antitumor drug potency in a human adenocarcinoma cell line” by Ruiz-Gómez MJ et al. of the Faculty of Medicine, University of Málaga, Spain. The resistance of tumor cells to antineoplastic agents is a major obstacle during cancer chemotherapy. The researchers explain: “Many authors have observed that some exposure protocols to pulsed electromagnetic fields (PEMF) can alter the efficacy of anticancer drugs; nevertheless, the observations are not clear”. They evaluated whether a group of PEMF pulses produces alterations of drug potency on a multidrug resistant human colon adenocarcinoma cell line, HCA-2/1(cch). The experiments were performed including:
a) Exposure to drug and PEMF for 1 h at the same time,
b) Drug exposure for 1 h, and followed by exposure to PEMF for the next 2 days (2 h/day).
Drugs used were vincristine, mitomycin C, and cisplatin. The cells were either treated with PEMF or sham exposed. Cell viability was measured by the neutral red stain cytotoxicity test. The results obtained were:
a) The 1 Hz PEMF increased vincristine cytotoxicity exhibiting 6.1% survival for which sham exposed groups showed a 19.8% of survival. Treatment with mitomycin C yielded a 5.3% survival rate as opposed to 19.2% in sham exposed group. Cisplatin showed a significant reduction in the % of survival from 44.2% to 39.1% in the sham exposed group.
b) Minor significant alterations were observed after nonsimultaneous exposure of cells to PEMF and drug.
They conclude: “The data indicate that PEMF can induce modulation of cytostatic agents in HCA-2/1(cch), with an increased effect when PEMF was applied at the same time as the drug. The type of drug, dose, frequency, and duration of PEMF exposure could influence this modulation”.
A study by Omote Y et al. of the Laboratory of Pathology, Hokkaido University School of Medicine, Sapporo, Japan, published in the Japanese Journal of Cancer Research in September 1990 investigates the “Treatment of experimental tumors with a combination of a pulsing magnetic field and an antitumor drug”. The effects of a combination treatment involving a pulsing magnetic field (PMF) and an antitumor drug, mitomycin C, on two experimental tumors (fibrosarcoma KMT-17 and hepatocellular carcinoma KDH-8) in WKA rats were studied. On day 7 after tumor implantation into the right thighs of rats, the region of the tumor was either exposed to PMF, or not, for 1 h immediately after iv injection of mitomycin C, or not. Survival rates at day 90 of KMT-17 implanted rats were 0% (0/10) in the non-treated group, 34% (4/12) in the mitomycin C-treated group, 47% (6/13) in the PMF-treated group and 77% (10/13) in the mitomycin C /PMF combination group. The increase of life span (ILS) of KDH-8-implanted rats in the combination therapy group was significantly prolonged (%ILS 17.6%) compared with that in the mitomycin C -treated (%ILS 3.4%) and PMF-treated (%ILS 7.6%) groups. By using cultured cells of the above two lines of tumor, the therapeutic effects of mitomycin C and PMF were also determined from the cell colony-forming efficiency in soft agar. The colony-forming efficiencies of both cell lines were significantly suppressed in the combination therapy group compared with those in the other single therapy groups. They conclude: “The present results indicate that PMF exhibited a potentiation of the antitumor effect of mitomycin C”.
Cancer Tumor and Hypoxia
A study published by the American Association for Cancer Research in 2004 entitled: “Daily Pulsed Electromagnetic Field (PEMF) therapy inhibits tumor angiogenesis via the hypoxia driven pathway: therapeutic implications” by Ivan L. Cameron, L. Z. Sun, N. Short, W. E. Hardman and C. D. Williams of the UT Health Science Ctr., San Antonio, TX, Pennington Biomedical Research Center, LSU, Baton Rouge, LA, EMF Therapeutics, Inc., Signal Mountain, TN, ten minute daily PEMF was found to retard angiogenesis and growth of a human breast cancer xenographt causing the tumor to develop proportionately larger areas of necrosis and hypoxia and smaller areas of proliferatively active cancer cells. It was also demonstrated that the daily PEMF therapy continued to inhibit tumor angiogenesis and tumor regrowth for two weeks following a standard course of ionizing radiation (IR) therapy, however this PEMF therapy renders larger areas of the tumor hypoxic therefore is expected to lessen susceptibility to oxidative damage caused by further IR treatments or by oxidative dependent chemotherapy. This leads to the conclusion that PEMF is an effective adjunct therapy following IR therapy but that the continued daily PEMF therapy should be stopped sometime (perhaps 2-4 days) prior to a second round of IR therapy. The temporary cessation of PEMF prior to the second round of IR is necessary for resumption of angiogenesis, decrease of hypoxic areas and increase in proliferative active and well oxygenated areas within the tumor for the second IR to be effective.
A later study by Cameron I.L. et al. published in Cancer Cell International in July 2005 confirms the effectiveness of PEMF in combination with gamma radiation in the control of tumor development in mice injected with human breast cancer xenograft. A variation of PEMF therapy was used in this study entitled “Therapeutic Electromagnetic Field (TEMF) and gamma irradiation on human breast cancer xenograft growth, angiogenesis and metastasis”. The mice were divided in four groups: a control group, a group treated with gamma radiation only, a group treated with TEMF only and a group treated with a combination of both gamma radiation and TEMF. Cameron et al. conclude: “Mice that received either IR or TEMF had significantly fewer lung metastatic sites and slower tumor growth than did untreated mice. No harmful side effects were attributed to TEMF. TEMF therapy provided a safe means for retarding tumor vascularization, growth and metastasis”.
Although no In Vivo human studies have been conducted thus far on the use of PEMF as adjunct therapy to the traditional cancer and tumor treatments, the many studies conducted In Vitro on tissue cultures and rodent In Vivo studies show great promise of obtaining better results with no known adverse side effects to date.
PEMF Therapy Increases Cellular Metabolism
In a study on Chronic Fatigue Syndrome and Electro-medicine, Thomas Valone, Ph.D, showed that damaged or diseased cells present an abnormally low TMP, about 80% lower than healthy cells. This signifies a greatly reduced metabolism and, in particular, impairment of the electrogenic Na+/ K+ pump activity associated with reduced ATP (Adenosine Tri-Phosphate) production.
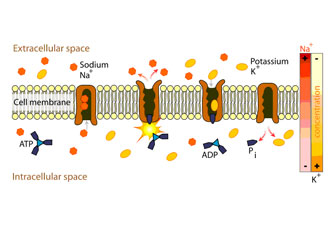 The Na+/ K+ pump within the membrane forces a ratio of 3Na+ ions out of the cell for every 2K+ ions pumped in for proper metabolism. The sodium-potassium pump uses energy derived from ATP to exchange sodium for potassium ions across the membrane. An impaired Na+/ K+ pump results in edema (cellular water accumulation) and a tendency toward fermentation,
The Na+/ K+ pump within the membrane forces a ratio of 3Na+ ions out of the cell for every 2K+ ions pumped in for proper metabolism. The sodium-potassium pump uses energy derived from ATP to exchange sodium for potassium ions across the membrane. An impaired Na+/ K+ pump results in edema (cellular water accumulation) and a tendency toward fermentation,
a condition known to be favorable toward cancerous activity.
French researcher Louis C. Kervran demonstrated that Sodium plus Oxygen plus Energy (ex: magnetic) nuclearly transmutes into Potassium as follows:
11 Na23 + 8 O16 + energy = 19 K39
This nuclear process is accomplished with low heat, in a low rate of thermal decomposition, which is the most important and commonly occurring phenomenon of Nuclear Fusion in Biology. As a result, utilization of oxygen in the cells increases and the body increases production of its own energy supplier (ATP). The organism becomes more stable and efficient; toxins and waste products are more rapidly broken down. The body's natural regulatory mechanisms are reinforced and healing processes accelerated.
Free radical proliferation is linked to pathological changes that cause cellular malfunction or mutation (i.e. cancer) as well as protein degradation. Free radicals also play a large role in causing damage to all cells of the body but particularly that of the immune system. According to studies, free radicals also “deplete cellular energy” by interfering with mitochondrial function and contribute to a shortened lifespan.
Cellular energy generation in the mitochondria is both a key source and a key target of oxidative stress in the cells. Seeking an electron to complete the radical, free radicals cause chain reactions as electrons are ripped from molecules, creating another free radical. Antioxidants such as vitamin A, vitamin E, selenium and coenzyme Q10 supply free electrons and are usually prescribed to provide limited relief in counteracting free radical ravages. However, electronic antioxidants produced by PEMF therapy can also satisfy and terminate free radicals by abundantly supplying the key ingredient usually found only in encapsulated antioxidant supplements…the electron (Thomas Valone, Ph.D. on Bioelectromagnetics, 2003).
On the biophysical level, as PEMF therapy increases the circulation of electrons across the cell membrane, a parallel phenomenon seems to occur, the acceleration of ATP synthesis and of other aspects of the cellular biochemical anabolism. As electrons are drawn to the inner membrane, they increase the ionic charge inside the cell and, thus, the TMP.
 In 1976, Nobel Prize winner Dr. Albert Szent-Gyorgi established that structured proteins behave like diodes or rectifiers. A diode passes electricity in only one direction. Dr. Szent-Gyorgi proposed that cell membranes can rectify an induced voltage and this rectifying property of cell membranes can cause changes in the ion concentration of the inner and outer surfaces of the cell membrane in such a way as to increase the TMP and effectively stimulate the activity of the Na+/ K+ pump. Cell health is directly affected by the health of the Na+/ K+ pump, which is directly proportional to the TMP.
In 1976, Nobel Prize winner Dr. Albert Szent-Gyorgi established that structured proteins behave like diodes or rectifiers. A diode passes electricity in only one direction. Dr. Szent-Gyorgi proposed that cell membranes can rectify an induced voltage and this rectifying property of cell membranes can cause changes in the ion concentration of the inner and outer surfaces of the cell membrane in such a way as to increase the TMP and effectively stimulate the activity of the Na+/ K+ pump. Cell health is directly affected by the health of the Na+/ K+ pump, which is directly proportional to the TMP.
Based on these biophysical principles, an endogenous high voltage EMF potential of sufficient strength will theoretically stimulate the TMP, normal cell metabolism, the sodium pump, ATP production and healing. Electro-medicine appears to connect to and recharge the storage battery of the TMP. Dr. Albert Szent-Gyorgi summarizes: “TMP is proportional to the activity of this pump and thus to the rate of healing.” Furthermore, “increases in the TMP have also been found to increase the uptake of amino acids.”
This is important, as increasing the supply of nutrients is also an effective aid to cell repair. This is particularly true in trauma where circulation has been impaired by crushed or severed blood vessels, or by the inflammation and swelling that compresses capillaries, blocking the flow to both the injured and uninjured cells.
PEMF Therapy Increases Energy Storage and Cellular Activity
At the sub-atomic level, as the pulsed fields expand and collapse through a tissue, the protein molecules, such as the cytochromes in the cells’ mitochondria, gain electrons and, in doing so, store energy. Even though the instantaneous peak magnetic energy amplitudes are very high, the average magnetic amplitudes generated by PEMF therapy remain low, the average total energy transmitted to the tissues is not powerful enough to create heat within the cells, nor for the cells’ atoms to vibrate much and cause a thermal increase, nor for an electron to jump to a higher orbit and emit heat as it returns to its orbit of origin. There is only sufficient average energy for the electron-spin to be increased, thus, energy gets stored in the cells’ mitochondria by converting ADP (Adenosine Di-Phosphate) to ATP molecules more rapidly by the addition of the phosphate
radical to the ADP. The ATP molecules store and transport the energy that is then used in the many chemical processes within the cell that participate in all the metabolic functions of living cells. This phenomenon is referred to as the electron transport chain and is described in the diagrams below.
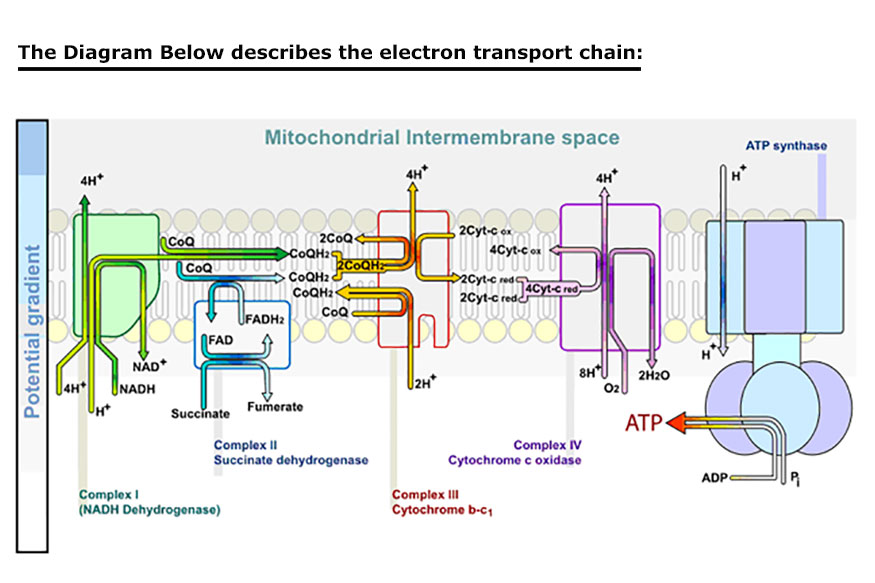
Understanding the effects of PEMF therapy at the atomic level requires a basic understanding of Quantum Mechanics that is provided here. Solving the Schrödinger equation for a molecule and determining probable amplitude for its electrons over an infinite number of possible trajectories yields the vibrational states of a molecule. This describes how the quantum state or wave function of a molecule or physical system changes in time. A diatomic molecule, which only involves one vibrational degree of freedom, (the stretching of the bond between the electron and the positron) provides a simple description (Atkins et al., 2002).
Quantum mechanical considerations show that during the electronic excitation of a particular molecule at the same orbital state, the energy of an excited triplet state (T1) is lower than that of its corresponding singlet state (S1). In biomolecules, the non-radiative crossing from the state S2 to S1 is generally the dominant mechanism. This crossing between two electronic states of the same spin multiplicity is called internal conversion (“IC”) (Atkins et al., 2002). The IC process is then followed by a rapid vibrational relaxation (decrease) where the excess vibrational energy is dissipated into heat, the molecule now ending up at the lowest, zero-point vibrational level of the S1 electronic state. From here, it can return to the ground electronic state S0 by emitting a photon (radiatively). The time-varying magnetic fields associated with PEMF therapy apparently affect electronic states via the intercrossing system (“ISC”), which is an excitation from state Si to Ti, where Ti is the corresponding triplet state (2 electrons are unpaired). The ISC type of crossing is heavily affected by the spin-orbit coupling, which relaxes the spin property by mixing with an orbital character (Szent-Gyorgyi A, 1976; Atkins et al., 2002). The ISC type of crossing leads to phosphorescence rather than fluorescence with radically different heat properties. Heavy metals, molecular oxygen having a triplet ground state, paramagnetic molecules such as hemoglobin, and heavy atoms such as iodine increase the inter-system crossing rate (Prasad, 2003).
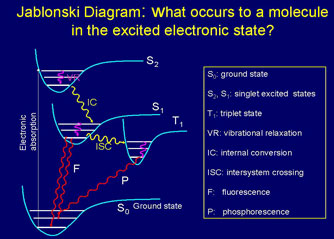
In shifting positions around an atomic nucleus, an electron generates energy and emits a magnetic resonance of specific frequency. Thus, the magnetic resonance field frequency of the various body tissues and organs is a product of the individual atomic, molecular and cellular frequencies specific to the molecules that constitute the particular tissue or organ. PEMF therapy therefore confuses the specific inherent magnetic resonance and temporarily modifies it in each atom, molecule, cell, and thus, tissue and organ.
From the perspective of biophysics, physiological markers represent a level of “order or disorder” in the magnetic resonance of a normal atom that correlates to internal and external factors. The Pulsed Electro-Magnetic Fields generated by PEMF therapy devices provide sufficient energy to affect the magnetic resonance of the atom as the electron is energized. When a disruption in the magnetic resonance occurs, the magnetic resonance of the electrons at the atomic level also exhibits a change, a phase shift that disturbs and breaks the once orderly pathways of communication that is usually transmitted from atom to molecule, molecule to cell, cell to tissue, and tissue to organ. In doing so, the phase shift influences the physical and chemical characteristics of the physiological markers.
PEMF therapy has proven beneficial in many ways for the various energetic body functions. All of the many types of living cells that make up the tissues and organs of the body are tiny electrochemical units. They are powered by a "battery” that is continually recharged by the cells' metabolic chemistry in a closed loop of biological energy.
PEMF Therapy Increases Cellular Membrane Flexibility and Elasticity
A study entitled “Modulation of collagen production in cultured fibroblasts by a low-frequency pulsed magnetic field” by Murray et al. (Biochim Biophys Acta) shows that the total protein synthesis was increased in confluent cells treated with a pulsed magnetic field for the last 24 h of culture as well as in cells treated for a total of 6 days. However, in 6 day-treated cultures, collagen accumulation was specifically enhanced as compared to total protein, whereas after short-term exposure, collagen production was increased only to the same extent as total protein. These results indicate that a pulsed magnetic field can specifically increase collagen production, the major differentiated function of fibroblasts, possibly by altering cyclic-AMP metabolism.
PEMF therapy successfully increases membrane flexibility by increasing the synthesis of collagen, a crucial protein that supports membrane elasticity, within the fibroblasts. In doing so, PEMF therapy increases tissue and muscle flexibility and, in doing so, increases range of motion.
PEMF Therapy Stimulates Cellular Communication and Replication
DNA synthesis is linked to pulsed, low intensity magnetic fields (Liboff et al., 1984; Rosch et al., 2004). Proteins are conductors of electricity. When exposed to strong fields, proteins are subject to electrophoresis. The Ribonucleic Acid (“RNA”) messengers that are synthesized from a Deoxyribonucleic Acid (“DNA”) template during transcription mediate the transfer of genetic information from the cell nucleus to ribosomes in the cytoplasm and serve as a template for protein synthesis. Since RNA mechanically influences the DNA and encoded proteins influence RNA, the flow of information to and from genes may be linked to changing magnetic fields (Einstein, 1977; Goodman et al., 1983).
Since magnetic fields interact with changing electrical charges and recent studies (Dandliker et al., 1997) show that DNA conducts electrons along the stacked bases within the DNA double helix, electro-magnetic fields may initiate transcription of the precursor mRNA by accelerating electrons moving within the DNA helix (McLean et al., 2003).
PEMF Therapy Increases Cellular Genesis (Cellular Growth and Repair)
The many intra and inter cellular processes and activity stimulated by PEMF therapy lead to faster cellular and tissue regeneration. This fact is shown by the results of many studies on a variety of tissues, including bones, spine, cartilage, intestines, blood vessels, nerves, brain, and muscles.
In December 2004, the Swiss Medical Tribune stated that PEMF therapy provided: “improvement of blood circulation, relief from pain, improvement of bone healing and the stimulation of nerve cells. Not only is the PEMF therapy effective in disease condition: it is an excellent means of preventing stress, assisting regeneration and recovery after sports exertion… Through metabolic activation and blood circulation more nutrients and oxygen are available to muscle cells, less damage is experienced, and efficiency is improved.”
PEMF and the Spine
In a long-term study entitled: “Spine fusion for discogenic low back pain: outcome in patients treated with or without pulsed electromagnetic field stimulation”, Marks RA. (Richardson Orthopedic Surgery, TX, USA) randomly selected 61 patients who underwent lumbar fusion surgeries for discogenic low back pain between 1987 and 1994 and had failed to respond to preoperative conservative treatments. Average follow-up time was 15.6 months postoperatively. Fusion succeeded in 97.6% of the 42 patients who received PEMF stimulation for only 52.6% of the 19 patients who did not receive electrical stimulation of any kind.
A similar study by Richard A. Silver, M.D. (Tucson Orthopedic & Fracture Surgery Associates, Ltd., Tucson, AZ, USA) with 85 patients who had undergone surgery of posterior lumbar inter body fusion (PLIF) and had risk factors associated with a poor prognosis for healing, including smoking, prior back surgery, multiple spinal levels fused, diabetes mellitus, and obesity, roentgen graphic examination and clinical evidence indicated that all but two patients achieved successful fusion. Of the 83 patients with successful spinal fusion, 29 (34.9%) were assessed as "excellent," 45 (54.2%) as "good," 3 (3.6%) as "fair", and 6 (7.2%) as "poor". Adjunctive treatment with PEMF appeared effective in promoting spinal fusion following PLIF procedures across all patient subgroups.
PEMF, Cartilage and Bones
In a study entitled: “Modification of biological behavior of cells by Pulsing Electro-magnetic fields”, 20 subjects of ages between 57 and 75 years with decreased bone mineral density as defined by a bone densitometer, were treated with PEMF therapy during a period of 12 weeks by Ben Philipson, Curatronic Ltd. (University of Hawaii School of Medicine, HI, USA). After a period of 6 weeks, the bone density rose in those patients with an average of 5.6%. Properly applied pulsed electromagnetic fields, if scaled for whole body use, have clear clinical benefits in the treatment of bone diseases and related pain, often caused by micro-fractures in vertebrae. In addition, joint pain caused by worn out cartilage layers can be treated successfully, through electromagnetic stimulation. PEMF application promotes bone union by electric current induction, which changes the permeability of cell membrane allowing more ions across, affects the activity of intracellular cyclic adenosine monophosphate (cAMP) and cyclic guano sine monophosphate (cGMP), and accelerates osteoblast differentiation by activation of p38 phosphorylation.
In healthy joints, movement generates electric currents in the joint. The electrical field in the joint permits the preservation and regeneration of the cartilage and surrounding bone and connective tissues (tendons, ligaments...). In the joint space, the chondrocytes produce cartilaginous extracellular matrix, creating space between individual cells. This matrix consists mostly of collagen type II and aggrecans. The aggrecan is a chondroitin sulfate proteoglycan that consists of two globular structural domains arranged into distinct regions, a chondroitin sulfate and a keratin sulfate region separated by a large domain modified with glycosaminoglycans.
 Keratin sulfates are large, highly hydrated molecules which act as a cushion to absorb mechanical shock. Glycosaminoglycans (GAGs) are highly negatively charged molecules with extended conformation that impart high viscosity to the solution. Along with the high viscosity of GAGs comes low compressibility, which makes these molecules ideal for a lubricating fluid in the joints. Injuries and osteoarthritis alter the electrical field in the joint and disrupt the normal mechanisms of preservation and regeneration of the cartilage.
Keratin sulfates are large, highly hydrated molecules which act as a cushion to absorb mechanical shock. Glycosaminoglycans (GAGs) are highly negatively charged molecules with extended conformation that impart high viscosity to the solution. Along with the high viscosity of GAGs comes low compressibility, which makes these molecules ideal for a lubricating fluid in the joints. Injuries and osteoarthritis alter the electrical field in the joint and disrupt the normal mechanisms of preservation and regeneration of the cartilage.
The ability of the cartilaginous tissue to withstand compressive stress is reduced. Once the destructive process has begun and external factors disturb the delicate integrity of healthy resting chondrocytes that are in maturational arrest, chondrocytes may undergo phenotypic dedifferentiation to a hypertrophic state. As the chondrocytes become hypertrophic, the mesenchymal cell layer moves further away, and distance between individual cells in the matrix becomes larger. The chondrocytes in the hypertrophic stage start to lose their cartilage-specific phenotype and produce a harmful substance, cartilage matrix degrading enzymes like MMPs and aggrecanases, in pathological quantities. This process hinders the production of proteoglycans, GAGs and chondroitin sulfate increasing the ratio of keratin to chondroitin sulfate.
The reduction in proteoglycans, the breaking up of the proteoglycan aggregates, their increased extractability and the change in hydration cause a reduction of the resistance of the amorphous substance and the collagen fibrils are subjected to intolerable mechanical pressure. The cavities created by enzymatic activity are subsequently filled by the hypertrophic chondrocyte with bone-specific collagen type I and X. Under the influence of vascularization from the underlying bone, the hypoxic state is released and eventually the chondrocytes become apoptotic and calcification of the matrix generates matured bone. As such, chondrocyte hypertrophy debilitates cartilage viability by a switched expression of bone-specific collagens, which initiates calcification of the matrix and up-regulation of cartilage-specific proteolytic enzymes and finally programmed chondrocyte cell death.
In a study entitled: “Effects of pulsed electromagnetic fields on patients' recovery after arthroscopic surgery: prospective, randomized and double-blind study” conducted at the "Sacro Cuore Don Calabria" Hospital, Negrar (Vr), Italy, and published in the Knee Surgery, Sports Traumatology, Arthroscopy journal in July 2007, Zorzi C et al. state: “Severe joint inflammation following trauma, arthroscopic surgery or infection can damage articular cartilage, thus every effort should be made to protect cartilage from the catabolic effects of pro-inflammatory cytokines and stimulate cartilage anabolic activities. Previous pre-clinical studies have shown that pulsed electromagnetic fields (PEMFs) can protect articular cartilage from such catabolic effects and prevent its degeneration resulting in chondroprotection. This prospective study was to evaluate the effects of PEMFs in patients undergoing arthroscopic treatment of knee cartilage. Patients with knee pain were recruited and treated by arthroscopy with chondroabrasion and/or perforations and/or radiofrequencies. They were randomized into two groups: a control group (magnetic field at 0.05 mT) and an active group (magnetic field of 1.5 mT). All patients used PEMFs for 90 days, 6 h per day. The patients were evaluated by the Knee injury and Osteoarthritis Outcome Score (KOOS) test before arthroscopy, and after 45 and 90 days. The use of non-steroidal anti-inflammatory drugs (NSAIDs) to control pain was also recorded. Patients were interviewed for the long-term outcome 3 years after arthroscopic surgery. Thirty-one patients completed the treatment. KOOS values at 45 and 90 days were higher in the active group and the difference was significant at 90 days (P < 0.05). The percentage of patients who used NSAIDs was 26% in the active group and 75% in the control group (P = 0.015).
At a three year follow-up, the number of patients who completely recovered was higher in the active group compared to the control group (P < 0.05). Treatment with PEMF aided patient recovery after arthroscopic surgery, reduced the use of NSAIDs, and also had a positive long-term effect.
Many studies demonstrate that PEMF stimulation protects cartilage from degeneration. PEMF increases blood and fluid flow, and in doing so, the partial oxygen pressure in the cells and the joint space and ion transport in the chondrocytes. This phenomenon helps maintain the chondroitin / keratin sulfate proteoglycan balance and levels of glycosaminoglycan in the matrix. Repair and growth of cartilage is stimulated, preventing further degradation and the grinding of the bones.
PEMF and Tendons
The Department of Rheumatology at Addenbrookes Hospital carried out investigations into the use of PEMF therapy for the treatment of persistent rotator cuff tendonitis. PEMF treatment was applied to patients who had symptoms refractory to steroid injection and other conventional treatments. At the end of the trial, 65% of these were symptom free, with 18% of the remainder being greatly improved.
In a study entitled “Pulsed Magnetic Field Therapy Increases Tensile Strength in a Rat Achilles’ Tendon Repair Model” published in 2006 (Department of Plastic and Reconstructive Surgery, Albert Einstein College of Medicine and Montefiore Medical Center, Bronx, NY; Department of Biomedical Engineering, Columbia University, New York, NY; and Department of Orthopedics, Mount Sinai School of Medicine, New York, NY.), Berish Strauch, MD. et al. conclude: “The use of electromagnetic fields in tissue healing is still a relatively recent application and much research remains to be performed. Areas that need greater explanation include the interplay between wound healing, contributing growth factors, and angiogenesis. PEMFs hold promise as a safe, easily administered and noninvasive modality to accelerate and improve the body’s healing mechanisms”.
PEMF and Intestines
An experimental study was designed to investigate the
effect of PEMF therapy on intestinal healing and to compare small and large intestinal anastomoses, or connections between the loops of the intestines, by Nayci et.al (Department of Pediatric Surgery, Mersin University Medical Faculty, Turkey). The study demonstrated that PEMF stimulation provided a significant gain in anastomotic healing in both small and large intestine, and a significant increase in both biochemical and mechanical parameters.
PEMF and the Brain
A four-week double-blind, placebo-controlled study conducted by Uni der Bundeswehr (Munich, Germany) assessed the efficacy of PEMF Therapy for Insomnia. One hundred one patients were randomly assigned to either active treatment (n = 50) or placebo (n = 51) and allocated to one of three diagnostic groups: sleep latency; interrupted sleep; or nightmares. The results showed 70% (n = 34) of the patients given active PEMF treatment experienced substantial or even complete relief of their complaints; 24% (n = 12) reported clear improvement; 6% (n = 3) noted a slight improvement. Only one placebo patient (2%) had very clear relief; 49% (n = 23) reported slight or clear improvement; and 49% (n = 23) saw no change in their symptoms. No adverse effects of treatment were reported.
Stunning results were obtained in a study entitled: “Protection against focal cerebral ischemia following exposure to a pulsed electro-magnetic field," Grant G et.al (1994 Department of Neurosurgery, Stanford University, CA, USA) stated: “There is evidence that electro-magnetic stimulation may accelerate the healing of tissue damage following ischemia. Exposure to pulsed electro-magnetic field attenuated cortical ischemia edema on MRI at the most anterior coronal level by 65%. On histological examination, PEMF exposure reduced ischemic neuronal damage in this same cortical area by 69% and by 43% in the striatum. Preliminary data suggest that exposure to a PEMF of short duration may have implications for the treatment of acute stroke”.
PEMF and Nerves
In 2003, Thomas J. Goodwin, Ph.D. of the NASA Lyndon B. Johnson Space Center, Houston, Texas, published a study on the “Physiological Effects of Time-Varying Electromagnetic Fields (TVEMF) on Human Neuronal Cells” based on model systems for growing two- and three-dimensional human neural progenitor cells”. “Neuronal tissue comprises elongated nerve cells composed of elongated axons, dendrites and nuclear areas. Axons and dendrites are chiefly responsible for transmission of neural signals over distance. Longitudinal cell orientation is critical for proper tissue formation and function, including growth”. The same electromagnetic field strength and all other conditions were identical for the two- (2D) and three-dimensional (3D) in vitro experimental conditions with the 3D cultures exposed to microgravity using a rotating wall vessel (RWV).
A study conducted by Lelkes and Unsworth in 1997 demonstrated that rotationally simulated microgravity has permitted some enhanced nerve growth. During the exposure to the TVEMF, the 2D cells were divided in two groups, a pool in direct contact with an electrode and pool not in direct contact with an electrode. All experimental and control groups were analyzed in triplicate and averaged for determination of mean and standard. The 2D pool cells exposed to a TVEMF displayed an accelerated growth rate and different morphology as compared to non-waveform cells (controls). The exposed cells preferentially aligned whereas the controls showed a normal random pattern. The cells in direct contact with the electrode remained stimulated up the 72 hours while the cells not in direct contact with the electrode continued to experience accelerated and long term stimulation in growth pattern even 168 hours after the TVEMF was removed. This would indicate that intense electric fields are not necessarily an essential component of this process, and that a weak and persistent stimulus might yield measurable effects. Viability was also higher in the cells exposed to the waveform. In each case, the cells returned to normal rates without further TVEMF stimulation. The 3D pool cells were monitored for cell proliferation, orientation, morphology, glucose metabolism and pH. The results showed little or no difference in metabolic parameters. The 3D RWV grown tissues emulated the same basic genetic response as the 2D tissues at similar fields’ strength.
The study showed that extremely low amplitude rapidly TVEMF of 10 to 200 mGauss (below the Earth’s field strength of 500 mGauss) exert a very potent effect on the proliferation, morphology and gene expression of the cells in both the 2D and 3D cultures as well as organized into 3D clusters in the RWV. Analysis of the gene array data yielded a significant down-regulation of about 175 maturation, differentiation and regulatory genes associated with the non-growth profile of normal cells and up-regulation of about 150 genes associated with growth and cellular proliferation. These genes are also primarily involved in the embryogenic process. The study successfully demonstrated the potential for TVEMF to control the proliferation rate, directional attitude and molecular genetic expression of normal neural progenitor cells, as well as control over the embryogenic development process. This electromagnetic potentiation can be used for a number of purposes including the development of tissues for transplantation, repairing traumatized tissues, moderating some neurodegenerative diseases and perhaps controlling the degeneration of tissue as might be effected in a bioelectric stasis field.
PEMF and Multiple Sclerosis
At the Biologic Effects of Light 1998 Symposium, Richards et al. explain the effects of pulsing magnetic field on brain electrical activity in multiple sclerosis: “Multiple sclerosis (MS) is a disease of the central nervous system. Clinical symptoms include central fatigue, impaired bladder control, muscle weakness, sensory deficits, impaired cognition, and others. The cause of MS is unknown, but from histologic, immunologic, and radiologic studies, we know that there are demyelinated brain lesions (visible on MRI) that contain immune cells such as macrophages and T-cells (visible on microscopic analysis of brain sections). Recently, a histologic study has also shown that widespread axonal damage occurs in MS along with demyelination. What is the connection between MS and bio-electromagnetic fields? We recently published a review entitled "Bio-electromagnetic applications for multiple sclerosis," which examined scientific studies that demonstrated the effects of electromagnetic fields on nerve regeneration, brain electrical activity (electro-encephalography), neurochemistry, and immune system components. These effects are important for disease pathology and clinical symptoms in MS”.
Richards referred to a study that evaluated electro-encephalograms (EEG) in response to photic stimulation with flashing lights before and after PEMF exposure. The evidence showed a increase in alpha EEG magnitude that was greater in the active group compared to the placebo group demonstrating increased activity. Richards et al. (Dep. Radiology, University of Washington, WA, USA) confirm the above conclusion in a double-blind study to measure the clinical and sub-clinical effects of an alternative medicine electromagnetic device on disease activity in multiple sclerosis. The MS patients were exposed to a magnetic pulsing device that was either active (PEMF) or inactive (placebo) for two months. Each MS patient received a set of tests to evaluate MS disease status before and after wearing the device. The tests included a clinical rating (Kurtzke, EDSS), patient reported performance scales (PS), and quantitative electro-encephalography (QEEG) during a language task. Although there was no change between pre-treatment and post-treatment in the EDSS scale, there was a significant improvement in the PS combined rating for bladder control, cognitive function, fatigue level, mobility, spasticity, and vision. There was also a significant change between pre-treatment and post-treatment in alpha EEG magnitude during the language task. Richards et al. stated: “we have demonstrated a statistically significant effect of the magnetic pulsing device on patient performance scales and on alpha EEG magnitude during a language task”.
In “Treatment with AC PEMFs normalizes the latency of the visual evoked response in a multiple sclerosis patient with optic atrophy”, Sandyk (1998, Department of Neuroscience at the Institute for Biomedical Engineering and Rehabilitation Services of Touro College, Dix Hills, NY, USA) explains: “Visual evoked response (VER) studies have been utilized as supportive information for the diagnosis of MS and may be useful in objectively monitoring the effects of various therapeutic modalities. Delayed latency of the VER, which reflects slowed impulse transmission in the optic pathways, is the most characteristic abnormality associated with the disease. Brief transcranial applications of AC PEMFs in the picotesla flux density are efficacious in the symptomatic treatment of MS and may also reestablish impulse transmission in the optic pathways… The rapid improvement in vision coupled with the normalization of the VER latency despite the presence of optic atrophy, which reflects chronic demyelization of the optic nerve, cannot be explained on the basis of partial or full reformation of myelin. It is proposed that in MS synaptic neurotransmitter deficiency is associated with the visual impairment and delayed VER latency following optic neuritis and that the recovery of the VER latency by treatment with PEMFs is related to enhancement of synaptic neurotransmitter functions in the retina and central optic pathways. Recovery of the VER latency in MS patients may have important implications with respect to the treatment of visual impairment and prevention of visual loss. Specifically, repeated applications of PEMFs may maintain impulse transmission in the optic nerve and thus potentially sustain its viability”.
Sandyk R. summarizes recent clinical work on the therapeutic effects of AC PEMF in MS: “Multiple sclerosis is the third most common cause of severe disability in patients between the ages of 15 and 50 years. The cause of the disease and its pathogenesis remain unknown. The last 20 years have seen only meager advances in the development of effective treatments for the disease. No specific treatment modality can cure the disease or alter its long-term course and eventual outcome. Moreover, there are no agents or treatments that will restore premorbid neuronal function. A host of biological phenomena associated with the disease involving interactions among genetic, environmental, immunologic, and hormonal factors, cannot be explained on the basis of demyelization alone and, therefore, require refocusing attention on alternative explanations, one of which implicates the pineal gland as pivotal. The pineal gland functions as a magneto-receptor organ. This biological property of the gland provided the impetus for the development of a novel and highly effective therapeutic modality, which involves transcranial applications of alternating current AC PEMFs flux density” (1997).
Summary
As demonstrated by the many studies cited herein, it is clear that PEMF treatment stimulates many aspects of cellular metabolism and activity by increasing the TMP and flow of ions across the cell membrane, growth factors, tissue repair and healing. PEMF therapy increases blood circulation in and around damaged tissue, and effectively helps damaged cells heal by bringing more oxygen into the cells. Effects that are observed when the TMP is increased include: enhanced cellular energy (ATP) production, increased oxygen uptake, changes in entry of calcium, movement of sodium out of the cell, movement of potassium into the cell, changes in enzyme and biochemical activity, and changes in cellular pH will stimulate large amounts of lymphatic vessels to pump and drain lymph fluid which, in turn, supports immune health. This effect involves a chain of processes in the human body, which leads to the improvement of health without side effects including:
- Increased production of nitric oxide
- Improved micro-circulation
- Increased supply of oxygen, ions and nutrients to cells
- Increased partial oxygen pressure
- Increased ATP production by excitation of electrons
- Stimulation of RNA and DNA production
- Accelerated protein bio-synthesis by electron and energy transfer
- Anti-oxidation regulation with increased circulation of available electrons
- Increased calcium transport and absorption for stronger bones, joints and muscles
- Enhanced cellular and tissue elasticity with increased collagen production
- Increased cellular genesis promoting bone, cartilage, tendon and soft tissue growth
- Stimulation of cellular repair mechanism
- Accelerated detoxification of cells and organs
- Increased absorption of nutrients and pharmaceuticals
- Enhanced macro circulation: by mechanically de-clumping blood cells, alternately dilating and constricting vessels, and through angiogenesis, the growth of new blood vessels
- Boosting of the immune system, the body's defenses, by improving the rolling and adhesion behavior of white blood cells
- Supporting the body's internal self-regulating mechanisms by activating cellular and molecular processes
- Decreased swelling, inflammation and pain
Beyond its complex mechanisms, PEMF therapy offers many health benefits. PEMFs help the natural body healing processes by delivering a non-invasive form of repetitive electrical stimulation that requires no direct contact with the skin surface. Magnetic fields have been shown to affect biologic processes and be effective in a wide range of medical conditions. PEMF therapy has proven beneficial in stimulating cellular metabolism, blood and fluids circulation, tissue regeneration and immune system response. Through these processes, cells are able to function better and tissues repair themselves more efficiently. Through the same processes, vital organs such as the liver, kidneys and colon are able to rid themselves of impurities thus detoxifying the body and allowing better organ functionality. PEMF treatment is effective at increasing bone formation and bone density, healing fractures and osteotomies, recovery from wounds and trauma, graft and post-surgical behavior, recovery from myocardial and brain ischemia (heart attack and stroke), tendonitis, osteoarthritis, and impaired neural function or spasticity from central nervous system diseases such as multiple sclerosis and spinal cord damage. PEMF stimulation offers a safer and more comfortable alternative for urinary incontinence to prior treatments. PEMF therapy improves sports performance, and simply helps to maintain good health. It stimulates muscles, connective tissues, intestines, tendons and cartilage, the brain and peripheral nerve sites. In doing so, PEMF therapy promotes healing and a return to higher activity levels. Functions that were lost begin to recover. Extensive research has been carried out to determine the mechanisms by which this occurs but, for the physiotherapist or other medical professional presented with a wide range of clinical problems, PEMF therapy is an invaluable aid to the clinic. PEMF therapy leaves you feeling relaxed, energized, renewed and with a sense of well-being.